|
This idea was much disputed at the time, and Louis De Broglie’s PhD thesis was almost not accepted. Einstein’s formula λ = h/mc would just need to be slightly rearranged into λ = h/mv, whereby m would be the mass of the particle, and v would be the velocity of the particle. A young French physics PhD student, Louis De Broglie had the radical idea that not only the photon, but all particles would exhibit the wave particle dualism, including the electron. The wave-particle dualism was originally thought to be valid for the photon only. Wave Particle Dualism of Massive Particles Figure 1.2.4 Max Planck, Nobel Prize 1918 (Attribution: /wiki/F.lanck_1933.jpg) As we will see later, not only electromagnetic radiation shows the wave particle dualism, but all particles including electrons. 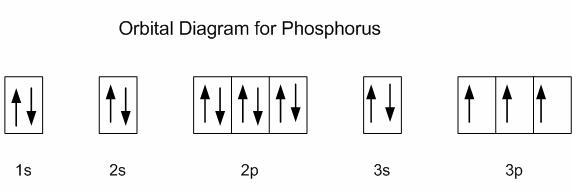
However, it is one of the most fundamental principles of nature. The discovery of the wave-particle dualism of electromagnetic radiation was a radically new concept that is difficult to grasp intellectually up to this date because the human mind tends to see waves and particles to be mutually exclusive. In fact, the mass of the particle associated with electromagnetic radiation, the photon, is inverse proportional to the wavelength of the electromagnetic radiation. This equation shows the wave particle-dualism of electromagnetic radiation because it relates a wavelength to a mass. With \(\nu\)=c/λ, and solved for λ, the equation becomes λ=h/mc. We can solve the equation for \(\nu\), and then it is \(\nu\)=mc 2/h. Because mass can be converted into electromagnetic radiation according to the equation E = mc 2, and the energy of electromagnetic radiation is E=h\(\nu\), mc 2=h\(\nu\). 
The wave-particle dualism of light, and electromagnetic radiation in general can also be mathematically derived. Only when the photon had an energy larger than the work function of the metal, the electron would be ejected and would have a kinetic energy equal to the difference between the energy of the photon and the binding energy. When light hits the metal surface the photon collides with the electron. Assuming photons the photoelectric effect could be easily explained (Figure 1.2.3, right). The quantization would be explained by the fact that light would not only have wave but also particle properties, and these particles would be called photons. This would mean that light was quantized. 1.2.4) based on the assumption that energy was quantized, and now Einstein had experimentally found it again in the quest to explain the photoelectric effect. The equation E=h\(\nu\) was previously derived by Planck (Fig. Figure 1.2.3 Maximum kinetic energy of the ejected electron as a function of the frequency of light (left), and mechanistic interpretation of the photoelectric effect (right). This would mean that electrons had an energy E=h\(\nu\) minus an energy E B that would be needed to overcome the binding energy, also called the work function, of the electron in the metal (Figure 1.2.3, left). He analyzed the slope of this line and found that the slope was the Planck constant h. He found that there was a linear relationship. Einstein investigated the maximum kinetic energy of the ejected electrons as a function of the frequency of the light. 
Below the threshold frequency no electrons get ejected. The threshold frequency depends on the metal. Above a certain frequency, or below a certain wavelength, light is able to eject electrons from the metal surface. The photoelectric effect occur when a metal surface is irradiated by light. 
, /wiki/F.lanck_1933.jpg) Figure 1.2.2 The photoelectric effect (Attribution: Wolfmankurd. Modified with Photoshop by PM_Poon and later by Dantadd. (Attribution: Photograph by Orren Jack Turner, Princeton, N.J. Figure 1.2.1 Albert Einstein, Nobel Prize 1921. It began with the investigation of the photoelectric effect by Albert Einstein (Fig. The phenomenon of the wave-particle dualism was first discovered for electromagnetic radiation, and the extended to all other particles including the electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
